Host Cell Protein (HCP)
Our team is uniquely skilled to offer first-class immunoassay development and analytical services for the detection and/or quantification of process-related contaminants, including host cell proteins.
Webinar
HCP Risk Management
Vincent Rivera, founder and director at GTP Immuno (part of GTP Bioways) is sharing his great experience regarding HCP (host cell proteins) and biocontaminants risk management.
Comprehensive services to validate your HCP ELISA kit
HCP risk management is involved at two critical development and manufacturing stages of your project:
- During process development and process validation to obtain relevant and specific data to improve downstream process development and further decrease HCP contamination
- During final product quality control
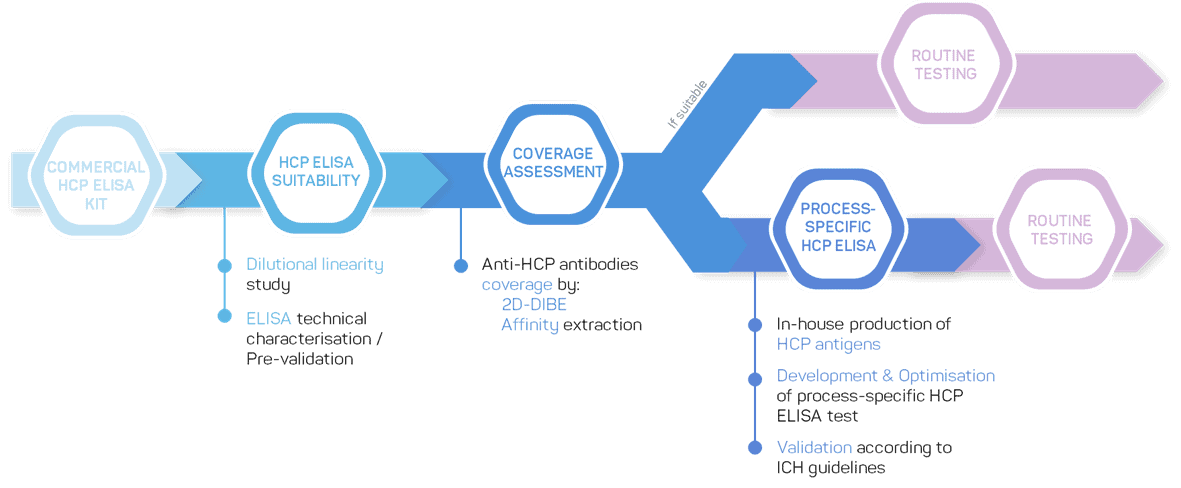
Prior to those steps, you will have to demonstrate the dilutional linearity of your HCP ELISA kit and assess the coverage of the antibodies for both process samples and the final biopharmaceutical product.
GTP Bioways has developed a strong expertise to assist you in the selection of the commercial HCP ELISA kit that best suits your project. If none of the commercial HCP ELISA kit fits your needs, our team can develop a process-specific HCP ELISA kit.

Process-specific HCP ELISA kit development and manufacturing
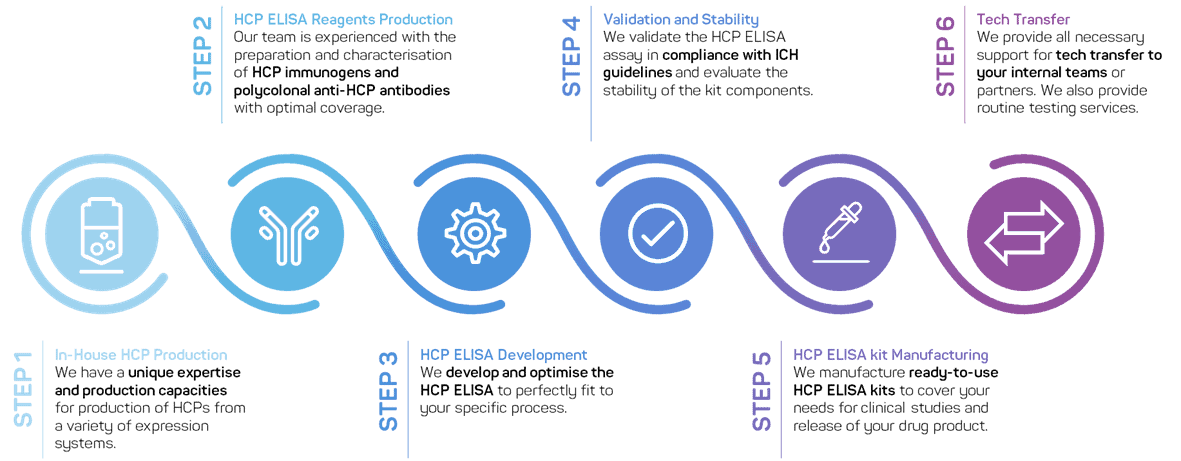
If no commercial kit is suitable for your project or when your project is ready to move to clinical phase, our team offers process-specific HCP ELISA development services:
GTP Bioways provides a wide range of analytical services dedicated to your biotherapeutics
We have the expertise to support the development of your biotherapeutics with our cutting-edge analytical & formulation services.
Analytical services
We offer expert analytical services dedicated to process development and GMP manufacturing.
Custom ELISA
We are experts in the development and manufacturing of custom immunoassays based on your requirements.
Formulation
Our seasoned team offers dedicated formulation development suitable to your specific biomolecule.

