About GTP Bioways
GTP Bioways is an expert and fast-growing contract development and manufacturing organization (CDMO) specialised in process development and manufacturing of antibodies, proteins, bioconjugates and nanodrugs.
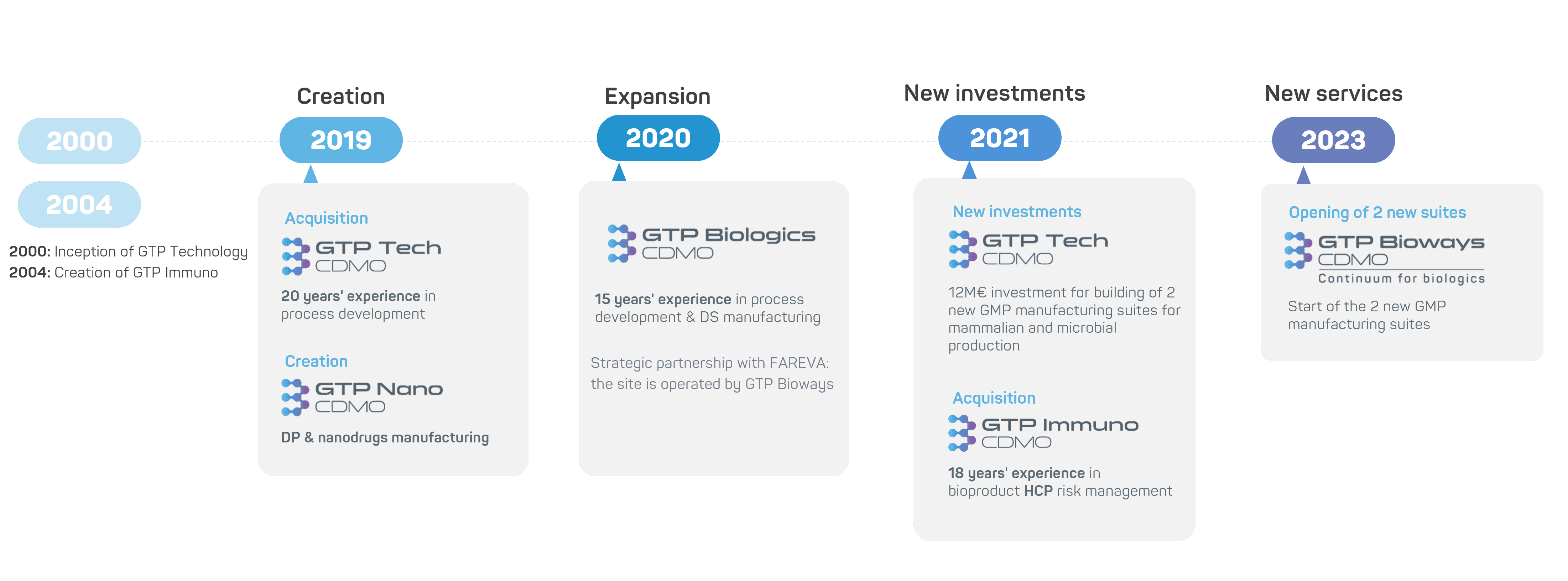
Our history
Today, with a team of almost 100 dedicated collaborators, GTP Bioways is focused on providing end-to-end services to support our customers at all stages of their drug development, from R&D to commercial manufacturing.
Expert & flexible CDMO for biologics, antibody conjugates and nanodrugs
GTP Bioways is a customer-centric and science-driven CDMO committed to offering uniquely customised process development and GMP manufacturing services for biologics, antibody-drug conjugates and nanodrugs.
With our cutting-edge facilities located in France, GTP Bioways can support biopharma companies from preclinical to commercial production of their innovative therapies.
Drawing on its expertise in solving difficult client problems, our team strives to offer a tailored approach, finely tuned to overcome your specific challenges. We are unique in our ability to develop agile solutions for challenging projects such as the production of difficult-to-express proteins, the development of atypical purification schemes or the formulation of nano-enabled products (NEP).


A science-driven and customer-centric CDMO
We are very proud of our teams of seasoned and highly qualified professionals and their extensive technical experience across all stages of the biopharmaceutical value chain, from R&D to cGMP production including aseptic filling.
Infusing a client-first corporate culture is a major priority of GTP Bioways’ management. We believe that empathy, integrity and transparency are key to ensuring long-lasting relationships with our customers and to the success of their programmes. We are proud to say that our customers praise our synergic approach.
Management team

Alain SAINSOT
President

Eric DEVIC
Managing Director

Damien TOURNIER
Chief Financial Officer

Hervé GINISTY
Chief Scientific Officer

Vincent RIVERA
Director of BU Immuno

Séverine HOUNTONDJI
Director of business development and marketing

Carine ARCENS
Corporate Quality Director


