GMP manufacturing of bioconjugates & ADC
Our state-of-the-art facilities and highly skilled team enable us to deliver a comprehensive range of services specifically tailored to your unique requirements for the manufacturing of antibody-drug conjugates (ADCs) and bioconjugates.
Dedicated GMP conjugation capabilities for cytotoxic payloads
Our advanced facilities are designed to meet the standards of major regulatory agencies. Our cGMP production suite is adapted for up to hundred-litre scale production of bioconjugates.
Our conjugation DS production suite is equipped with:
- Our isolator technology which ensures utmost safety in the handling of highly cytotoxic substances (OEB6).
- A stainless steel bioreactor for conjugation
- Single-use purification equipment (chromatography and TFF)
Along with well-established controls for handling cytotoxic materials (highly potent APIs & extremely potent APIs), we have implemented a quality system and documentation process in compliance with CFR guidelines.

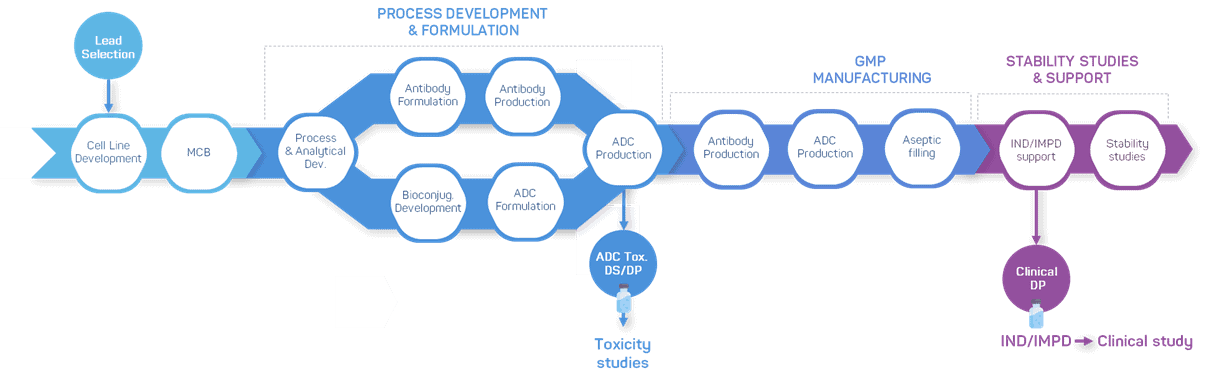
Streamline your ADC production with our comprehensive development & manufacturing services
With our unmatched expertise and capabilities, we seamlessly cover every stage of your antibody drug conjugate project, from antibody intermediate production to bioconjugation and GMP manufacturing of drug substance and drug product.
Comprehensive analytical package for bioconjugates characterisation
With over a decade of expertise, our analytical development team is committed to guiding you through every step of your bioconjugate development. Bioconjugates, such as antibody drug conjugates (ADCs), present unique challenges due to their complex composition.
Our expertise lies in tailoring analytical methods specifically to each moieties within the bioconjugate, ensuring comprehensive characterisation. From protein/mAb intermediates to the final bioconjugates, our comprehensive analytical methods package covers a wide range of techniques to support your development. Specifically tailored for ADCs, our analytical package includes, but is not limited to:

- Drug Antibody Ratio (DAR) and quantification of residual free drug
- Fragment profile, size exclusion chromatography
- Micro-Flow Imaging
- Cytotoxicity
- Residual solvent, if any solvent is used during the manufacturing process
- Mass spectrometry, used for characterisation and batch release
Our experienced analytical team has the right expertise to handle method transfer, method development, and validation – ensuring a robust process for the successful cGMP manufacturing of your bioconjugate drug substance and drug product.



