CHO Cell line development
GTP Bioways offers SMART cell line development services to provide you with highly producing and stable mammalian cell lines ready for transfer to GMP manufacturing.
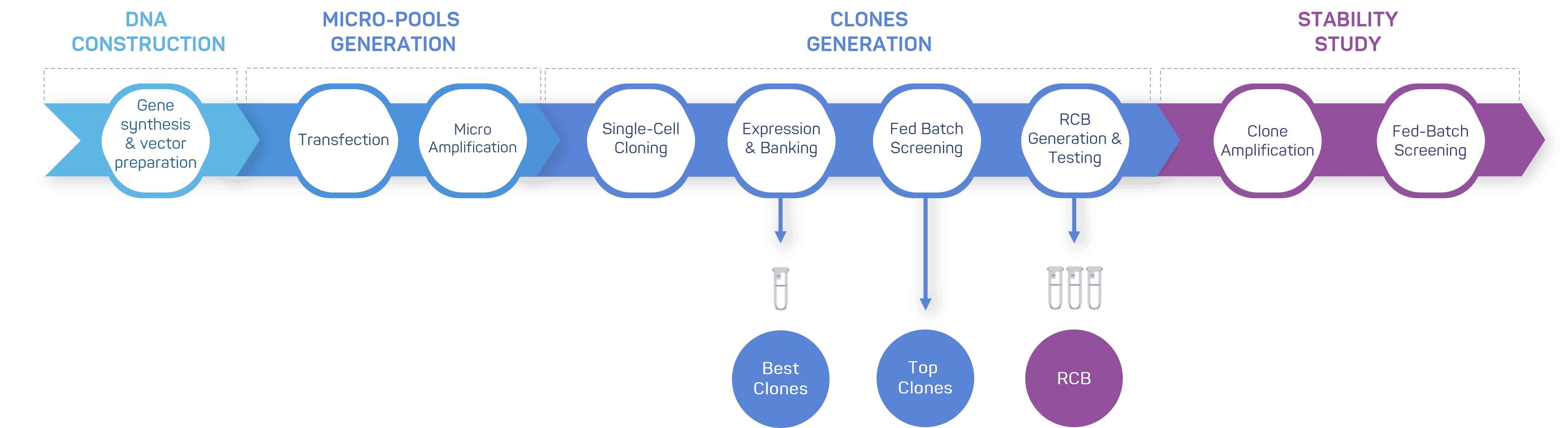
Efficient workflow for accelerated CHO cell line development
With a well-established workflow for the cloning, transfection and selection of the top clones, we are able to offer competitive timelines.
In addition to providing an increased assurance of clonality, this process means we can, starting by the generation of micro-pools, offer an early preparation of non-GMP materials and accelerate process development for projects with strict time constraints.


Efficient and recognised CHO platform
GTP Bioways’ cell line development services rely on the CHOvolution™ technology, an established, robust and highly performant CHO platform.
The technology is based on a non-genetically modified CHO-K1 host cell line with a proprietary vector system which ensures the insertion of at least two copies of the expression cassette into the genome. Together with the use of a strong promoter, this guarantees a strong expression of the gene of interest.
Frequently Asked Questions (FAQ) about Cell Line Development

What are the main cell lines used for stable production of antibodies?
The cell lines commonly used for stable production of antibodies are:
- Chinese Hamster Ovary (CHO) cells: CHO cells are the most widely used cells for industrial production of therapeutic antibodies. They offer advantages such as high protein expression levels, compatibility with mammalian systems, and the ability to perform proper post-translational modifications like glycosylation.
- NS0 cells: These murine myeloma cell lines are often used to produce monoclonal antibodies. They have the capability to produce high levels of antibodies and are commonly used in hybridoma technology, where the antibody-producing hybridoma cells are generated by fusing B cells with myeloma cells.
- Human Embryonic Kidney (HEK) 293 cells: HEK 293 cells are human-derived cell lines that are widely used for transient expression of antibodies and other recombinant proteins. They offer high transfection efficiency and the ability to produce large quantities of proteins within a short period. HEK 293 cells offer the advantage of achieving closer glycosylations compared to CHO cells. However, they are less commonly used for stable antibody production since HEK cells are more sensitive to process parameters (g. temperature, stirring, and shear forces).
It is important to note that the choice of cell line depends on various factors, including the specific requirements of the antibody, desired post-translational modifications, scalability, and regulatory considerations. However, CHO cells remains the gold standard of biopharmaceutical production.
What are the advantages of CHO cells ?
CHO (Chinese Hamster Ovary) cells have several advantages that make them a popular choice for biopharmaceutical production:
- Expression Capability: CHO cells are a robust expression system, capable of producing high levels of recombinant proteins, including complex proteins such as antibodies, enzymes, hormones, and growth factors.
- Scalability: CHO cells can be easily scaled up from small laboratory cultures to large-scale bioreactors (high robustness and good resistance to shear stress), allowing for efficient and cost-effective production of recombinant proteins.
- Regulatory Approval: CHO cell lines have a long history of successful use in the biopharmaceutical production, and their safety and performance are well-documented.
- Industry Standard: CHO cells have become the industry standard for biopharmaceutical production due to their proven track record, established protocols, and extensive knowledge base. This makes it easier to find expertise, resources, and raw material specifically tailored for their use for the manufacturing of a biologic.
How long does it take to generate a stable cell line for antibody production?
The timeline for generating a stable cell line for antibody production can vary depending on several factors, including the complexity of the antibody, the specific cell line engineering techniques employed, and the efficiency of the selection process.
Thanks to our well-established workflow for cloning, transfection, and selection of top clones, we are proud to offer competitive timelines for generating stable cell lines. For standard monoclonal antibodies (hIgG), we have streamlined our process to efficiently generate Research Cell Banks (RCBs) within a timeline of just 3 months from DNA.
What is your process for developing a stable CHO cell line expressing a mAb?
Our process for cell line generation begins with the co-transfection of two expression vectors, each containing the gene of interest (GOI) for the protein. Upon transfection, we apply a selection pressure to ensure that only cells that have integrated the vectors into their genome are selected.
Cells are then grown to a certain level before a limiting dilution is performed. After an incubation period, thousands of supernatants are collected and subjected to an ELISA analysis. This analysis aims to identify cell lines with the highest productivity. From this initial transfection pool, we select the top clonal cell lines.
The productivity of the best-performing cell lines is evaluated by fed batch production and we select the top clonal cell lines that exhibit the highest productivities and allow to obtain a product with the required quality attributes. Research Cell Banks (RCBs) are generated for each of these top clones and a stability study is performed.
Which selection criteria do you apply to select the most promising clones in your cell line development service ?
Following the single-cell cloning step, our selection process focuses on identifying the top 40 clones from a transfection pool of approximately 2000. In this initial selection, the criteria are solely based on productivity, including volumetric productivity and specific productivity of the desired protein.
However, after the fed-batch screening, our selection approach shifts to include additional factors. In this phase, we carefully monitor metabolites consumption and growth markers of the candidate cell lines. By evaluating these parameters, we gain insights into the overall performance and potential of the cell lines beyond productivity alone. This comprehensive assessment allows us to make informed decisions regarding the most promising clones for further advancement in the cell line development process
Which antibody formats can you produce?
Our cell line development service offers the capability to produce virtually any recombinant protein including:
– Full-length antibodies of different isotypes
– Antibody fragments (Fab, scFv, diabody…)
– Bispecific antibodies
– Antibodies fused to other proteins
What are the unique advantages of your stable cell line generation service ?
Our team has almost 10 years’ experience in cell line development for a diversity of molecules including standard monoclonal antibodies (mAbs), complex antibody formats, and other classes of recombinant proteins.
We understand that some proteins require customized approaches, and we are used to adapt our platform process to meet your specific project requirements. For instance, we can develop or optimize specific ELISA protocols to screen clones not only on productivity but also on activity, or to screen for clones expressing bispecifics.
Our expertise in the field enables us to provide key features as part of our cell line development service:
- To maximize the likelihood of inserting the gene of interest into the cell genome and enhance cell expression, we employ the co-transfection of two expression vectors.
- Following the limiting-dilution step, we have the capability to screen and analyse thousands of growing cell lines. This extensive screening process ensures to obtain several clonal cell lines with the ability to overexpress your protein of interest.
- Five hyper-producers are selected at the end of our cell line development program and tested for their stability to produce your protein of interest over a large number of generations. This allows us to make sure that the cell line will be suitable for large-scale fed-batch productions.
What type of regulatory guarantees do you provide regarding your cell line development service?
In the realm of biopharmaceutical development, cell line development holds tremendous importance. At GTP Bioways, we recognize the criticality of this activity and ensure full traceability to support the filling of regulatory documents.
All operations performed during the process, including the use of consumables and starting genetic material, are thoroughly documented. This comprehensive documentation allows for complete traceability and provides a reliable history of the cell line, vector, and plasmid employed in constructing the final Research Cell Bank (RCB).
In addition, proof of clonality (which is required by regulatory authorities) of your cell line is ensured by combining single-cell printing in 96-well plates and kinetic whole-well imaging.

